How Heat Treatment Cycles Alter the Molecular Structure of Steel: Lessons from Everyday Nail Clippers
Steel is one of the most versatile materials in engineering, and its properties are dramatically transformed through heat treatment cycles. These controlled heating and cooling processes rearrange the molecular (crystalline) structure of iron-carbon alloys, turning soft, ductile steel into hard, wear-resistant material — or vice versa.
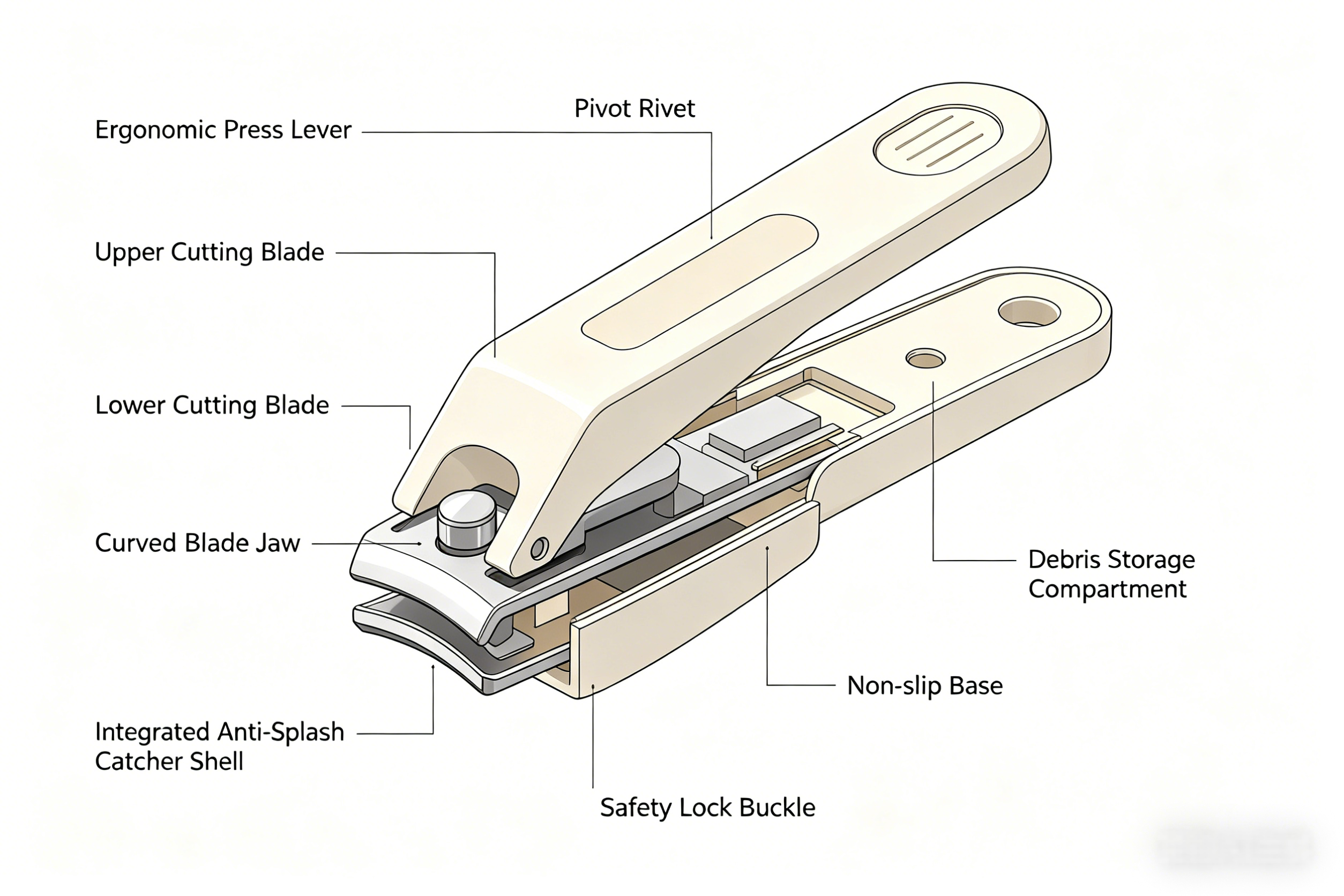
A perfect everyday example is the humble nail clipper. The blades must be hard enough to cut nails cleanly without dulling quickly, yet tough enough to avoid chipping. Manufacturers rely on precise heat treatment to achieve this balance.
Understanding Steel’s Molecular Structure
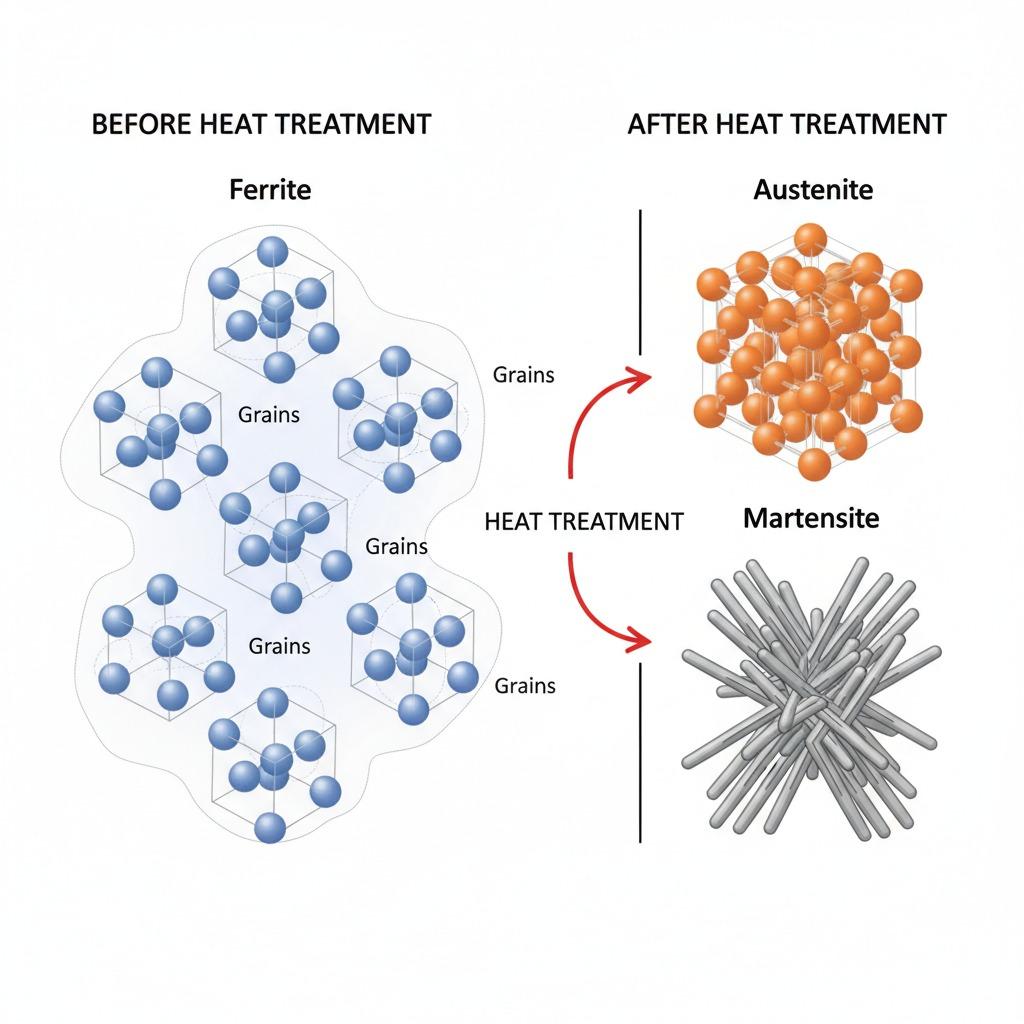
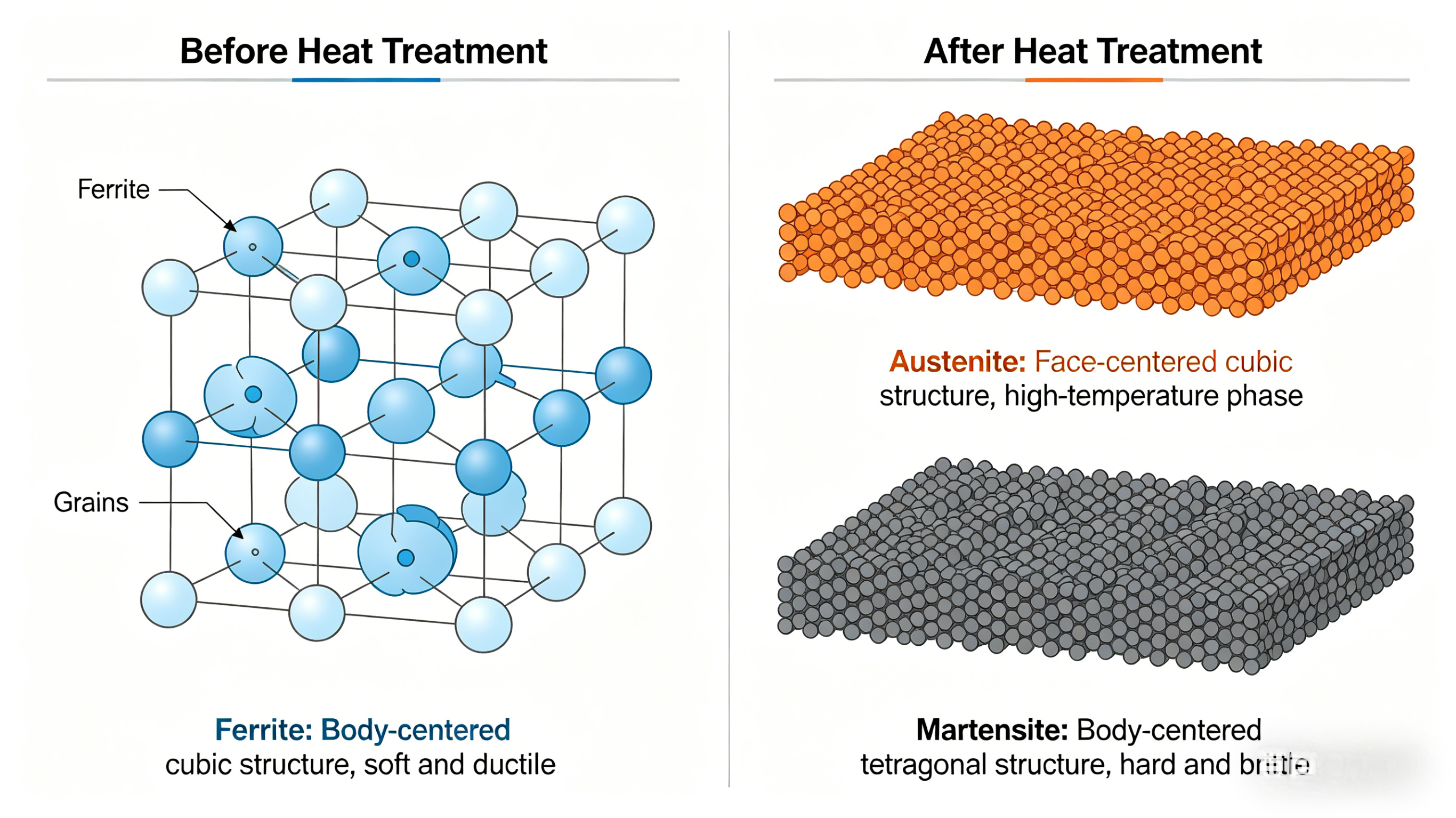
Steel’s base structure is iron with dissolved carbon. At room temperature, it typically consists of:
- Ferrite (body-centered cubic, soft and ductile)
- Pearlite (lamellar mixture of ferrite and cementite)
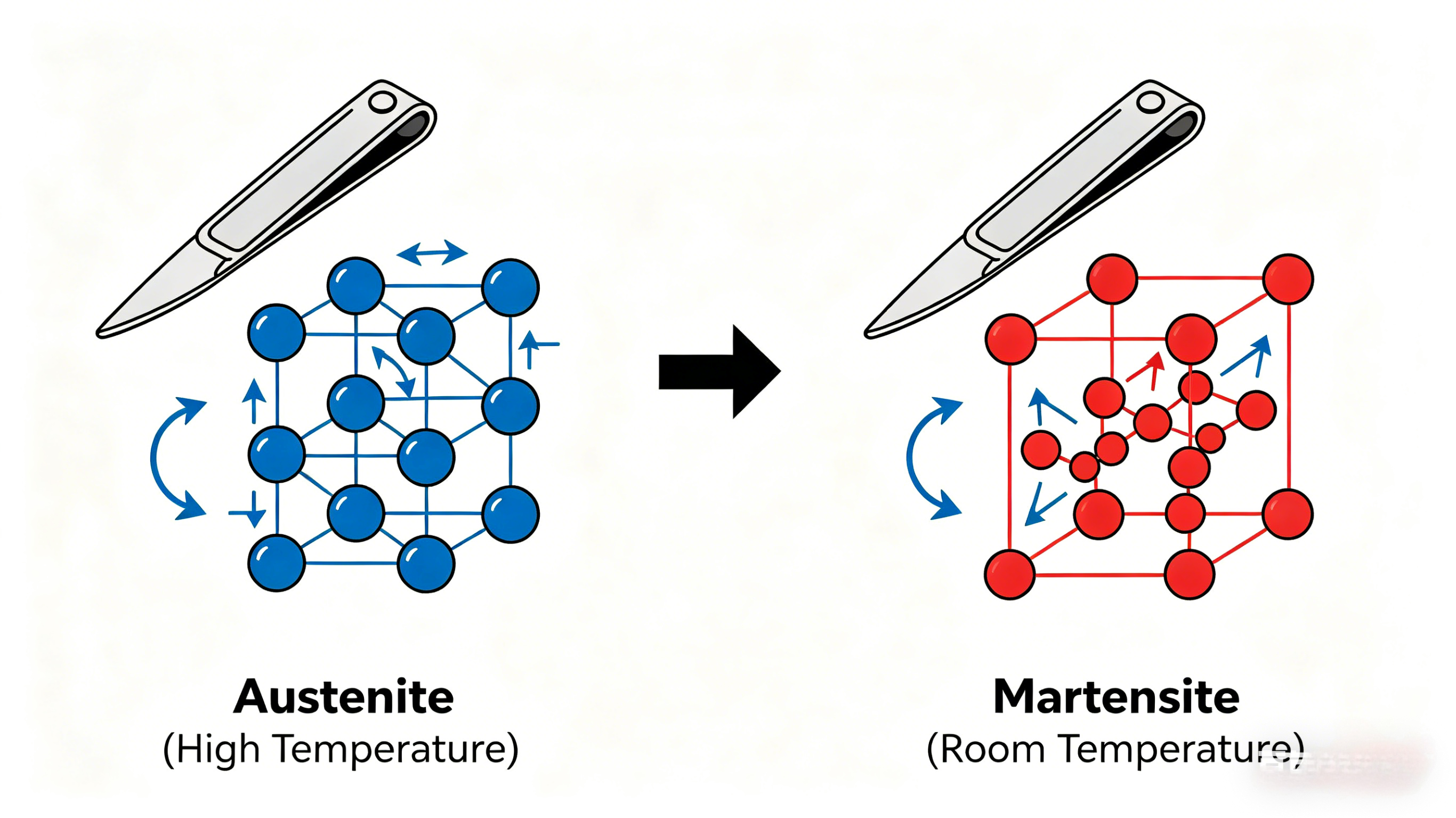
- Austenite (face-centered cubic, stable at high temperatures)
- Martensite (body-centered tetragonal, hard and brittle when formed rapidly)
Heat treatment manipulates phase transformations by controlling temperature, time, and cooling rates.
Common Heat Treatment Cycles and Their Effects
Here are the primary processes:
- Annealing Heat to ~700–900°C (above critical temperature), hold, then slow furnace cooling. Result: Coarse pearlite + ferrite → soft, machinable steel with low internal stress.
- Normalizing Heat to austenitizing temperature, air cool. Result: Finer pearlite → uniform structure, better toughness than annealed steel.
- Quenching Rapid cooling (oil, water, or polymer) from austenite region. Result: Martensite formation → very hard but brittle.
- Tempering Reheat quenched steel to 150–650°C. Result: Martensite partially transforms into tempered martensite → balances hardness and toughness.
Nail clipper blades typically undergo quenching followed by tempering to achieve high hardness (Rockwell C 50–60) while retaining enough ductility.
Comparison Table of Heat Treatment Effects
| Process | Heating Temp | Cooling Method | Primary Microstructure | Hardness | Ductility/Toughness | Typical Nail Clipper Application |
|---|---|---|---|---|---|---|
| Annealing | 700–900°C | Very slow (furnace) | Coarse pearlite + ferrite | Low | High | Not used for blades |
| Normalizing | 850–950°C | Air cool | Fine pearlite | Medium | Medium-High | Structural parts |
| Quenching | 800–900°C | Rapid (oil/water) | Martensite | Very High | Low | Initial blade hardening |
| Tempering | 150–650°C | Air cool | Tempered martensite | High | Medium-High | Final step for nail clipper blades |
| Austempering | 800–900°C | Salt bath ~300°C | Bainite | High | High | Premium durable tools |
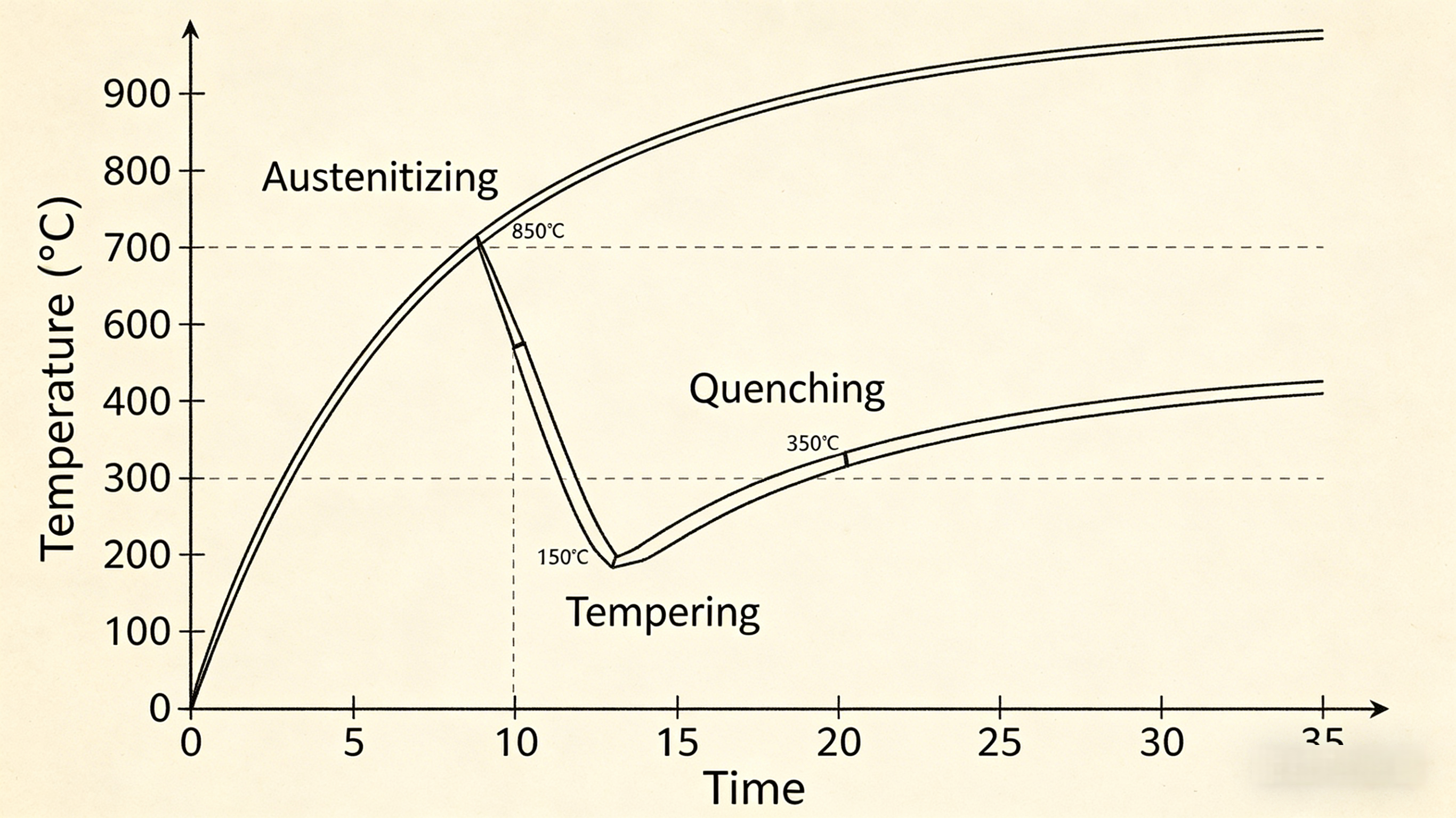
Visualizing the Heat Treatment Cycle for Nail Clippers
Typical cycle for high-quality steel nail clippers:
- Austenitize at ~850°C
- Quench in oil to form martensite
- Temper at ~300–400°C to relieve brittleness
- Optional surface treatments or sharpening
This sequence gives the blades the perfect combination of edge retention and shock resistance.
Why Molecular Changes Matter
The transformation from austenite to martensite involves a sudden shear rearrangement of carbon atoms trapped in the iron lattice, creating immense internal stresses that make the material hard. Tempering allows some carbon to precipitate as fine carbides, reducing brittleness while preserving strength.
Without proper heat treatment, a nail clipper would either bend too easily or snap under pressure.
Conclusion
Heat treatment is both science and art — turning ordinary steel into high-performance components we use daily. Next time you reach for your nail clipper, remember the sophisticated molecular dance its blades underwent in the factory furnace.
Understanding these cycles helps engineers design better tools, from surgical instruments to automotive parts. Whether you're a metallurgist, maker, or simply curious about everyday objects, the story of steel reveals how temperature and time can rewrite a material’s destiny at the atomic level.